The 25.2 releases of the LORENZ RIM solution are here: drugTrack, eValidator, and docuBridge
Posted on October 31, 2025
Release Highlights:

What's new in LORENZ RIM? The User Notification Center now offers improved preference settings, including an Opt-Out per Category option, giving you full control over which notifications you receive. The updated Best Practice Configuration has also been refined based on customer feedback and real-world experience, making it easier for new users to get started and for existing ones to benefit from smoother setup processes. Available for both cloud and on-premise customers, it now includes broader Assisted Compilation coverage, overall usability improvements, and simplified upgrade options requiring less manual effort. Discover more about these features here: What's new in RIM 25.2?
This entry was posted in LORENZ Solutions and Services.
Sneak peek: Four Ways our 25.2 release will make your regulatory life easier
Posted on October 27, 2025

Sneak peek at a 25.2 RIM feature:
This entry was posted in LORENZ Solutions and Services.
The 25.1 releases of docuBridge, drugTrack, and eValidator are available!
Posted on April 30, 2025
Release Highlights:

What's new in docuBridge? The advanced Node Content Pane improves efficiency in complex eCTD 4.0 applications with faster performance, clearer replace overviews, and customizable document views. The new webAccess App lets you use docuBridge in a distraction-free environment on Chrome or Edge, with all the same features as the browser version. The webAccess Explorer now includes Search Management, so authorized users can create, edit, share, and manage searches and application views with ease. Discover more about these features here: What's new in docuBridge 25.1?
This entry was posted in LORENZ Solutions and Services.
Sneak peek at our upcoming 25.1 releases
Posted on April 25, 2025
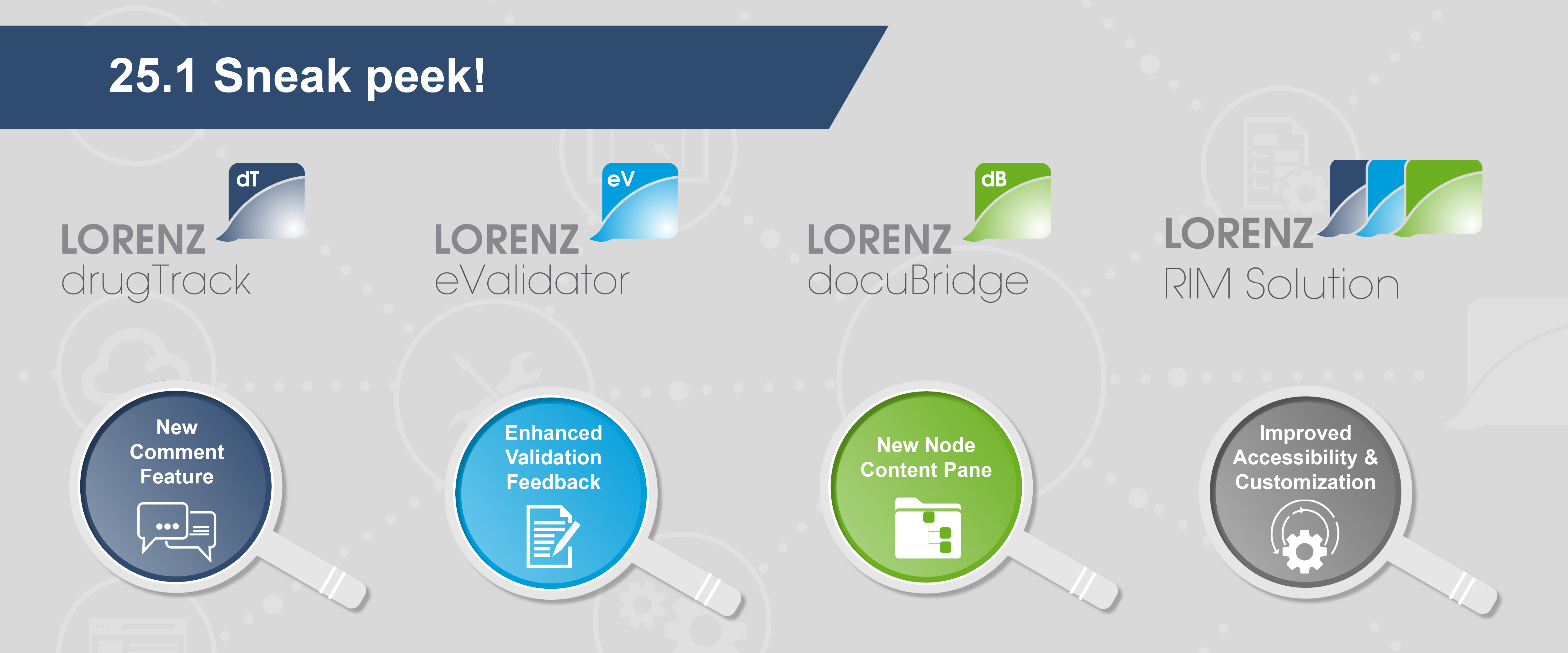
Sneak peek at the docuBridge 25.1 release:
This entry was posted in LORENZ Solutions and Services.
The 24.2 releases of docuBridge, drugTrack, and eValidator are available!
Posted on October 31, 2024
Release Highlights:

What's new in docuBridge? Among the latest enhancements we make a big step towards eCTD 4.0 adoption and introduce an exciting development. With seamless eCTD 4.0 forward compatibility, you can now transition smoothly from eCTD v3.2 to v4.0, ensuring efficient, future-ready submissions. The Submission Repository has also been upgraded with structured pools, a new "Packages" location type, and improved workflows, offering more flexibility and control over file management. Plus, the new Audit Trail Timeline provides a clear, filterable view of all document changes, enhancing transparency and oversight. Discover more about these features here: What's new in docuBridge 24.2?
This entry was posted in LORENZ Solutions and Services.
Sneak peek at our upcoming 24.2 releases
Posted on October 24, 2024

Sneak peek at the docuBridge 24.2 release:
This entry was posted in LORENZ Solutions and Services.
The 24.1 releases of docuBridge, drugTrack, and eValidator are available!
Posted on April 30, 2024
Release Highlights:

What's new in docuBridge? Unlock enhanced productivity with docuBridge's latest release! Seamlessly collaborate with our User Notification Center, receiving instant updates on document annotations and interactions. Stay engaged with notifications triggered by note modifications, fostering teamwork and streamlining communication. Effortlessly organize your workspace with multiple content panes in webAccess, enhancing workflow efficiency. Simplify PDF comparison in the Submission Workbench, saving time and ensuring accuracy in document analysis. Click here to read more about these highlights: What's new in docuBridge 24.1?
This entry was posted in LORENZ Solutions and Services.
Sneak peek at our upcoming 24.1 releases
Posted on April 24, 2024

Sneak peek at the docuBridge 24.1 release:
This entry was posted in LORENZ Solutions and Services.

